A Duke University Medical Center phase two study will soon be looking for 100 adults who recently suffered a stroke caused by a blood clot to test the efficacy of unrelated cord blood as a treatment. As this is a placebo-controlled study, participants will receive either a single dose of cord blood or a placebo. Transfusions will be performed three to 10 days following the stroke, and researchers will then follow the individuals for one year to determine the safety and effectiveness of the treatment.
Phase Ones results
This is a continuation of a 2015 phase one study that demonstrated cord blood treatments to be feasible and well-tolerated with improved functionality for the patient. In that study, all 10 males between 45 and 79 years of age showed improvements of one to three grade levels within three months of receiving cord blood. This is encouraging because only 58% of stroke patients show improvements in their grade levels within three months, according to other studies.
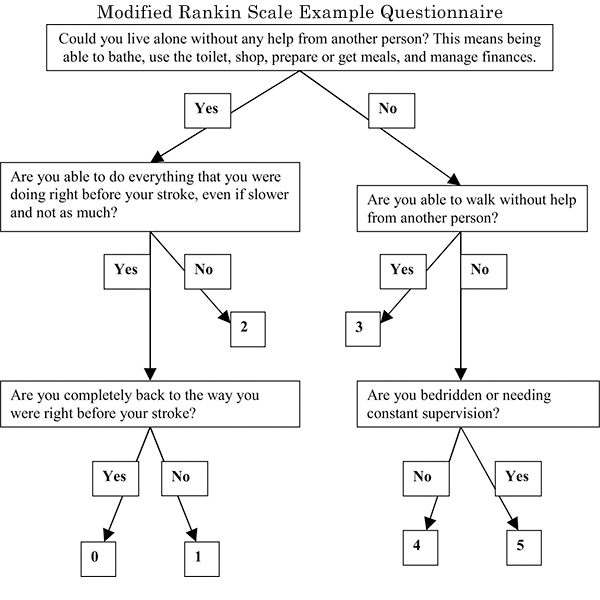
Rankin Scale
Grade levels are on the modified Rankin Scale, which ranges from zero to six. The scale is a simple, high-level analysis. For instance, people who can look after their own affairs but say they are unable to carry out all previous activities receive a grade level of two, which denotes a slight disability. People who are unable to attend to their own bodily needs without assistance, on the other hand, will receive a grade level of four, which indicates a moderately severe disability.
NIH stroke scale
Another way researchers measured the results was through the National Institutes of Health Stroke Scale (NIHSS). It comprises 11 tests measuring sensory, motor, cognitive, language and other functions and skills. Participants in the phase one study showed improvements of four to nine points, out of a total of 42 points, within three months on the NIHSS scale. A single point on the NIHSS scale could mean the difference between the patient being able to answer a simple two part question correctly such as providing both the current month and his or her current age or being able to perform two tasks together like blinking while squeezing his or her hands.
These clinical trials follow the results of a phase one study at Stanford that left its researchers “stunned” by the improvement they saw in the motor function of its participants.
Candidates for this new trial must be 18–90 years of age and meet other eligibility criteria.